Researchers Have Created Nanosprings To Break Down Microplastics
Dhir Acharya - Aug 15, 2019
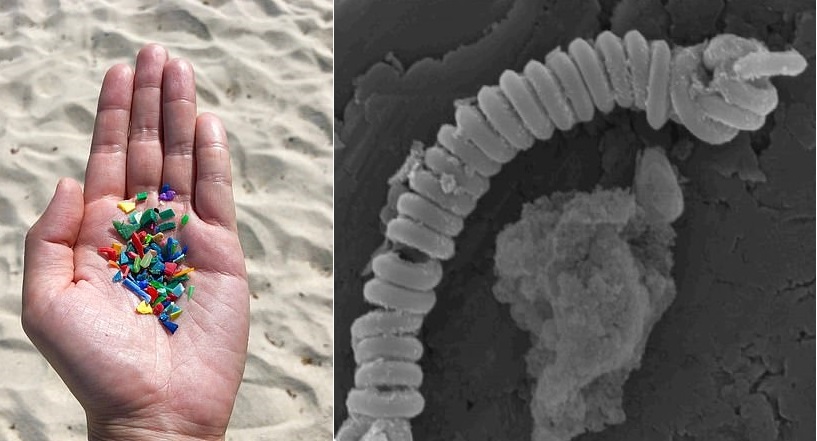
Engineers have created carbon nanosprings that break down microplastics, which pollutes rivers and oceans, without harmful impacts on microorganisms.
- Surprise! You're Eating 50,000 Microplastic Particles A Year, How Does That Feel?
- The Shocking Truth: All Sea Turtles Are Suffering From MicroPlastics Inside Their Bodies
Engineers from China and Australia have created carbon nanosprings, giving hope for breaking down microplastics, which pollutes rivers and oceans, without harmful impacts on microorganisms.
In addition, the engineers have designed these tiny springs to become magnetic after use, which means we can recollect and reuse them.
The project, described in the journal Matter, brought together several researchers from the Guangdong University of Technology in China, Australia’s Edith Cowan University, Curtin University, and the University of Adelaide.
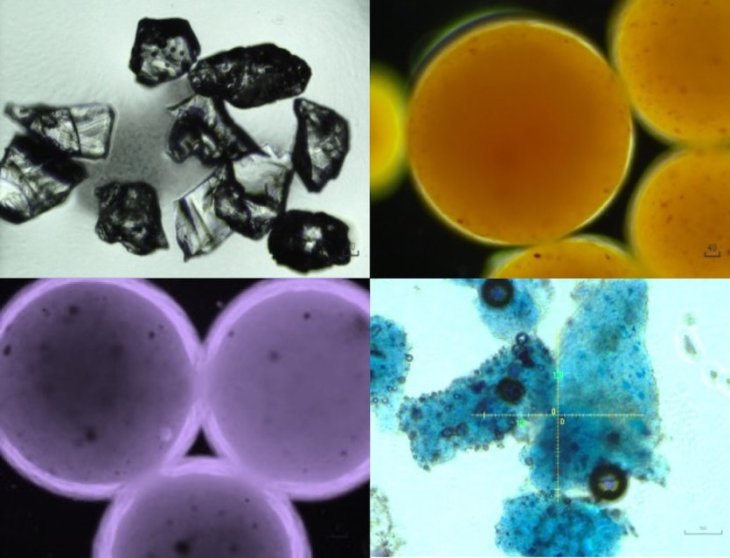
While we cannot see microplastics with our naked eyes, these are very common pollutants. Some of them are just so small that we cannot filter out in industrial water treatment, for example, the exfoliating beads existing in cosmetics. Others come from larger debris such as soda tires and bottles as they weather.
In their latest research, the team created reactive oxygen species, the short-lived chemicals that trigger chain reactions to chop long molecules (which make up microplastics) into harmless, tiny segments that will dissolve in the water.
However, generally, we need heavy metals like cobalt or iron to produce reactive oxygen species. These metals are dangerous pollutants and can’t be used for the sake of the environment. So, the team used carbon nanotubes laced with nitrogen for boosting the production of reactive oxygen species.
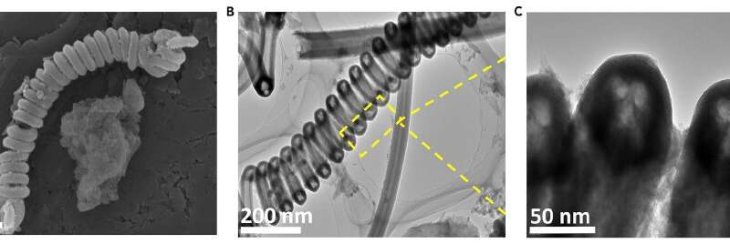
The catalysts for the carbon nanotube removed microplastics significantly within only eight hours, according to researchers. Meanwhile, they are still stable in the harsh oxidative conditions required to break down microplastics.
And with the coiled shape, the stability is increased while the reactive surface area is maximized. And when the researchers include a small quantity of manganese far from the nanotubes’ surface so that it doesn’t leach into the water, the tiny springs became magnetic.
Xiaoguang Duan, a co-author from the University of Adelaide said:


Because each microplastics has different chemical properties, the next step for the team is to make sure the nanosprings is effective with microplastics of different shapes, origins, and compositions.
In addition, the researchers want to continue to confirm that any chemical compounds that occur as by-products or intermediates during microplastics decomposition is non-toxic.
The team also hopes that they can harness these by-products as a source of energy for microorganisms plagued by polluting plastics.
As per senior author Sahobin Wang from Adelaide:

Featured Stories

Features - Mar 24, 2026
How to Use GeForce NOW to Play Video Games Without Actual Hardware

Features - Jan 29, 2026
Permanently Deleting Your Instagram Account: A Complete Step-by-Step Tutorial

Features - Jul 01, 2025
What Are The Fastest Passenger Vehicles Ever Created?

Features - Jun 25, 2025
Japan Hydrogen Breakthrough: Scientists Crack the Clean Energy Code with...

ICT News - Jun 25, 2025
AI Intimidation Tactics: CEOs Turn Flawed Technology Into Employee Fear Machine

Review - Jun 25, 2025
Windows 11 Problems: Is Microsoft's "Best" OS Actually Getting Worse?

Features - Jun 22, 2025
Telegram Founder Pavel Durov Plans to Split $14 Billion Fortune Among 106 Children

ICT News - Jun 22, 2025
Neuralink Telepathy Chip Enables Quadriplegic Rob Greiner to Control Games with...

Features - Jun 21, 2025
This Over $100 Bottle Has Nothing But Fresh Air Inside

Features - Jun 18, 2025
Comments
Sort by Newest | Popular